-
Meat withdrawn from French supermarkets over E.Coli risk
Lidl and Super U among stores selling potentially impacted ground beef
-
Ryanair becomes most popular carrier at Toulouse airport
Several low-cost carriers are targeting the French city with route expansions
-
New reports of Britons missing flights due to EES delays
Queues of several hours reported in Spain prior to full EES rollout
French health body takes action to prevent Paracetamol shortage
New rules limit the number of boxes per person bought without a prescription. Authorities say supplies are ‘under tension’
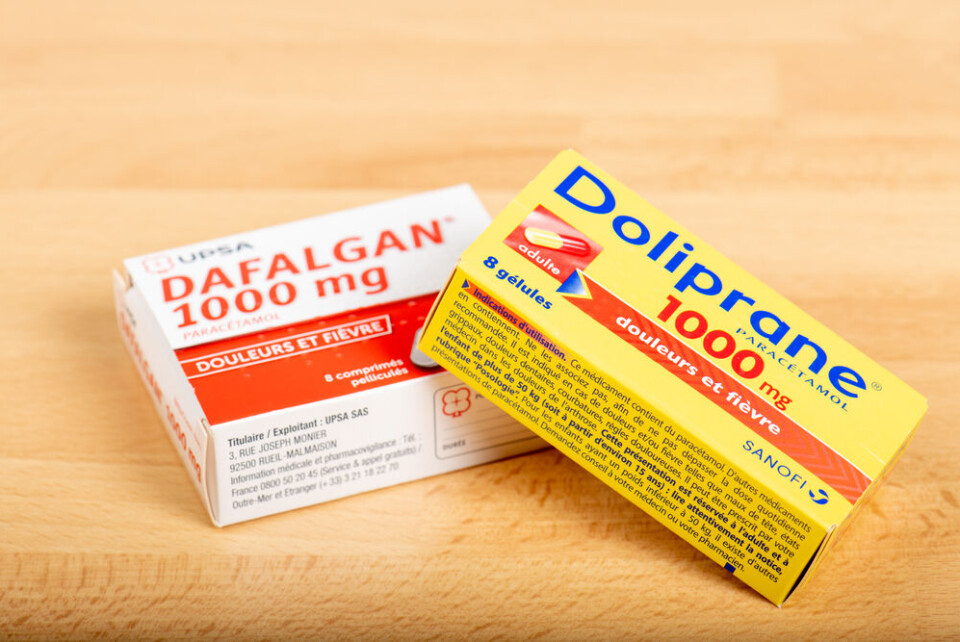
Health authorities in France have brought in new measures to prevent a shortage of paracetamol, including limits on its purchase.
Read more:France faces summer paracetamol shortages due to supply delays
Medicine safety agency l'Agence nationale de sécurité du médicament (ANSM) has advised pharmacies to:
- Prioritise prescriptions for paracetamol-based medicines
- Limit the purchase of paracetamol to two boxes only for people without a prescription
Brands affected include Doliprane, Dafalgan, and Efferalgan.
Read more:What are the French equivalents of some common UK medicine brands?
On its website, the ANSM said: “Currently, there are delivery delays for the oral form and suppository form of paracetamol.”
Mélanie Cachet, director of the ANSM inspection department, told AFP that the measures had been brought in place to prevent a shortage of paracetamol rather than because stocks are at risk now.
She said: “[ANSM] has ensured that supplies are distributed fairly throughout the country and that available stocks are maintained over time.”
Wholesalers are also prohibited from exporting the medicine. However, the ANSM said it expects the situation to return to normal after the summer.
Ms Cachet said: “These [temporary] measures will allow all patients to have access to the medicine.”
Hospital supply has also been assured, she said.
Drug manufacturing lab Sanofi has also said that “there is no risk of a lack of paracetamol in France” and said that “all of our teams are working at the maximum to respond best to patient needs”.
Upsa, which packages more than 200 million boxes per year in its Agen factory (of which 115 million are destined for the French market), told Le Figaro: “We can guarantee continued access to our medicines. There is no backlog of orders and we are following the situation closely.”
Philippe Besset, president of the pharmacist union le syndicat de pharmaciens FSPF (Fédération des syndicats pharmaceutiques de France), said: “We have had high demand since winter, due to Covid and flu, so stocks have not built back up.”
However, he said that paracetamol was “under tension” rather than “out of stock”.
Shortages of paracetamol have been at risk since 2020, after the first wave of Covid, the ANSM said, when measures to protect supply were also introduced.
This is because the drug was recommended as an at-home remedy for people who caught the virus, as opposed to ibuprofen, which the public was advised against.
























