-
Blood thinner recalled in France over contamination fears
Health agency says nationwide recall 'precautionary measure'
-
19 inspirational French women for International Women’s Day
Samantha David takes a look at the life and work of women she admires - who would you add to the list?
-
People are living longer in better health in France, official new data shows
The expected healthspan of people in France has increased by almost two years since 2008
40,000 women in France have potentially faulty contraceptive coils
The affected coils have reduced contraceptive benefits and could lead to unwanted pregnancy. They do not pose an urgent health risk but should be removed
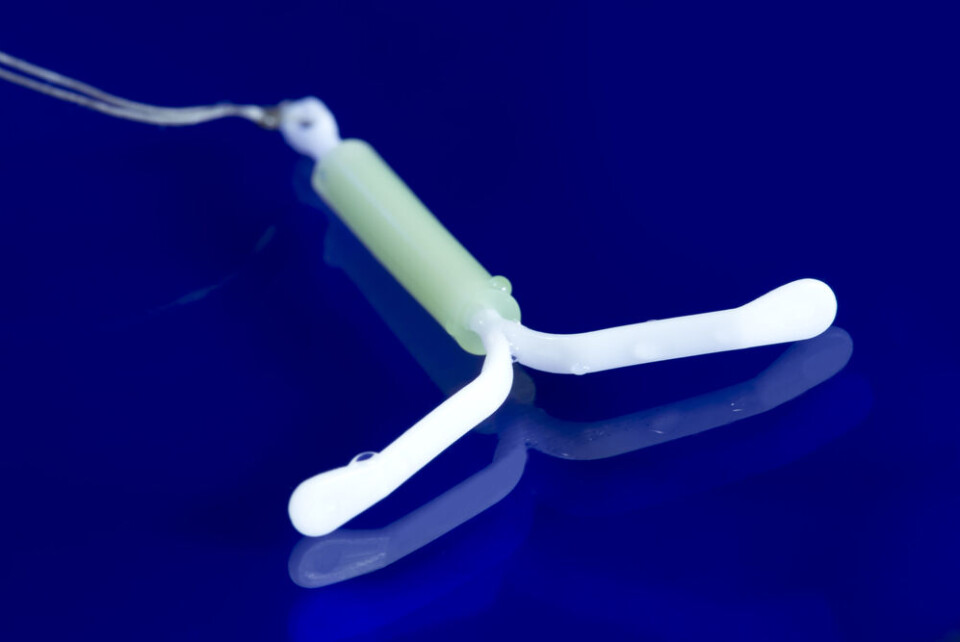
Certain contraceptive coils should be removed in “non-urgent” procedures due to the risk of unexpected expulsion, the French medicines agency (ANSM) has said.
The advice concerns coils (also called intrauterine devices or IUDs) made by the brands Ancora and Novaplus, which are thought to be used by up to 40,000 women in France.
Women using the coils most likely had them fitted from 2017 to March 2019.
Sales of affected coils made by the two brands have been suspended since 2019. Both are made by Spanish producer Eurogine.
Thierry Thomas, deputy director of ANSM, said that removing the coils was “a measure of precaution not urgence, to be discussed in the next meeting with a gynaecologist”.
Women affected could also discuss removal with their GP or midwife, he added.
Signs that there is an issue with the affected coils – such as abdominal pain, bleeding outside periods, IUD strings being too long or disappearing and pain during sex – should be discussed with a medical practitioner as soon as possible.
The main risk of using the defective coils is loss of contraceptive benefits leading to unwanted pregnancy.
A recommendation to remove the coils after three years of use was previously made by ANSM, as most unexpected expulsion occurred after this point. This advice has now been updated.
Up until 2017, products from Eurogine made up 5% of coil sales in France, being used by around 20,000 women per year. In 2018, this fell to 3%.
Related stories
French start-up installs urinals for women in Paris
You can add married name to French Covid vaccine certificate, says EU
























