-
Storm warning: 44 French departments placed on alert
Dramatic change in weather expected this weekend following sunny spell
-
Private medical laboratories to strike from May 4
Industrial action will not impact hospital labs but may delay non-urgent tests
-
TotalEnergies extends fuel price cap and announces special offer on diesel in France
Drivers can expect to pay a maximum of €1.99 per litre for petrol and €2.25 per litre for diesel this month
Covid antibody drug authorised for immunodeficient in France
People in France who are at risk of severe forms of the virus even after full vaccination will now be eligible to receive the drug Ronapreve as a preventive measure
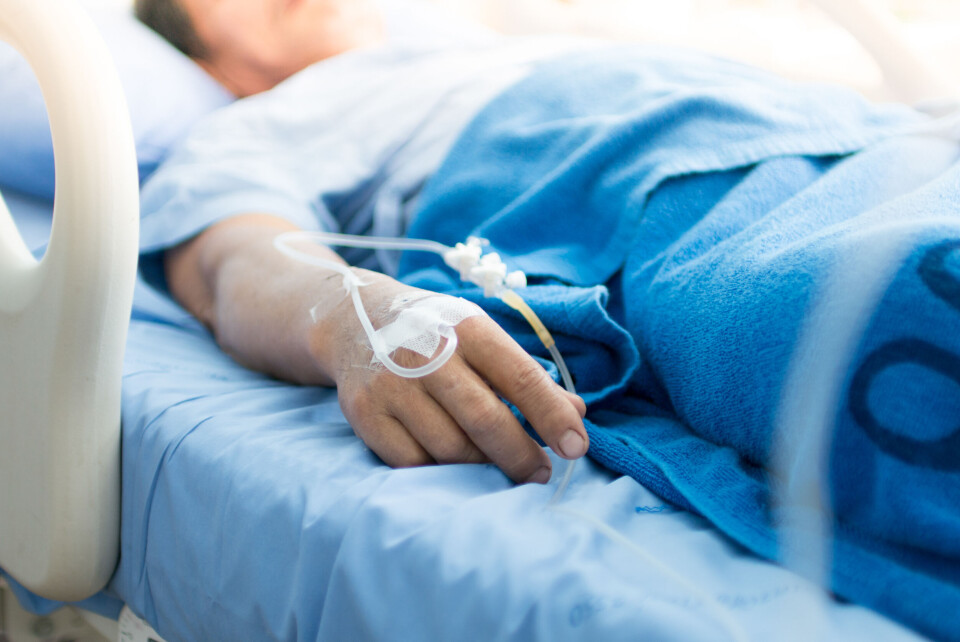
Immunosuppressed people who have low or no antibodies against Covid-19 even after full vaccination will now have access to ‘urgent’ preventative treatment, the French health authority has confirmed.
These people are at ‘very high risk’ of developing a severe form of Covid once they are exposed to the virus, especially in light of the spread of the Delta variant, the Haute Authorité de Santé (HAS) said.
In a statement, it said it was authorising “early access” to preventative treatment for immunosuppressed or immunodeficient patients, including those who are identified as contacts of infected people.
A proportion of these patients have no antibody protection against Covid-19 even after full vaccination, as a result of their condition, the HAS said.
In general, immunosuppressed patients have a reduced ability to fight infections and other diseases, because of other conditions such as AIDS, certain genetic conditions or because they are undergoing treatment for other illnesses, such as chemotherapy for cancer.
The HAS has now said that these patients will have priority access to preventative treatment, named ‘Ronapreve’, by Roche-Regeneron, which brings together the two monoclonal antibodies, casirivimab and imdevimab.
It said: “Around 25% of immunodeficient people, for example after a transplant or chronic lymphocytic leukaemia, still have no detectable antibodies [in their blood] even after a third dose” of the vaccine.
These patients will benefit from the Ronapreve treatment “either as prevention, or after exposure to SARS-CoV-2 [the virus that causes Covid-19]”, the HAS said, meaning if they are identified as a Covid contact case.
Around 130,000 people are “non-responsive to a full vaccination”, and are eligible for the treatment, it said.
It recommended that a national register should be compiled of patients who are treated with the drug, in order to “evaluate the biological and clinical effectiveness of the treatment”.
Data will need to be collected every two months, the HAS said, and the "decision whether to renew this authorisation will take place six months from today”.
What is Ronapreve?
Ronapreve is produced by American biotech company Regeneron, in partnership with pharmaceutical giant Roche.
It has also been approved as a Covid treatment in the UK, and was used to treat former US President Donald Trump when he contracted the virus.
It is given by injection or drip, and administers the artificial antibodies casirivimab and imdevimab. These work specifically against the coronavirus S ‘spike’ protein, which would otherwise enable the virus to enter cells.
The UK has authorised Ronapreve’s use as a Covid treatment but said that it does not constitute “a replacement” for vaccination. In contrast to France, the UK has so far not listed who will be eligible for the drug.
Experts in the UK have said that it should be reserved for the most vulnerable, as it costs £1,000-£2,000 (€1,166-€2,332) per use.
Related stories
France hits 40 million goal for full vaccinations against Covid-19
Covid France: Pressure mounts on hospitals; most patients unvaccinated
EU orders promising treatment for severe Covid-19 from UK pharma firm























