-
New map shows where ticks are prevalent in France
People spending time outdoors are advised to ‘be prepared’ against the disease-carrying parasites
-
Chance to win a €1m Picasso with a €100 ticket in Paris raffle
Tickets are now on sale ahead of the draw later this month
-
British snowboarder found dead in French Alps after cliff fall
43-year old man fell 100 metres while snowboarding off-piste
How close is France to finding a vaccine to treat cancer?
A French firm will unveil positive results of its first clinical trial this week at a key cancer congress in the US
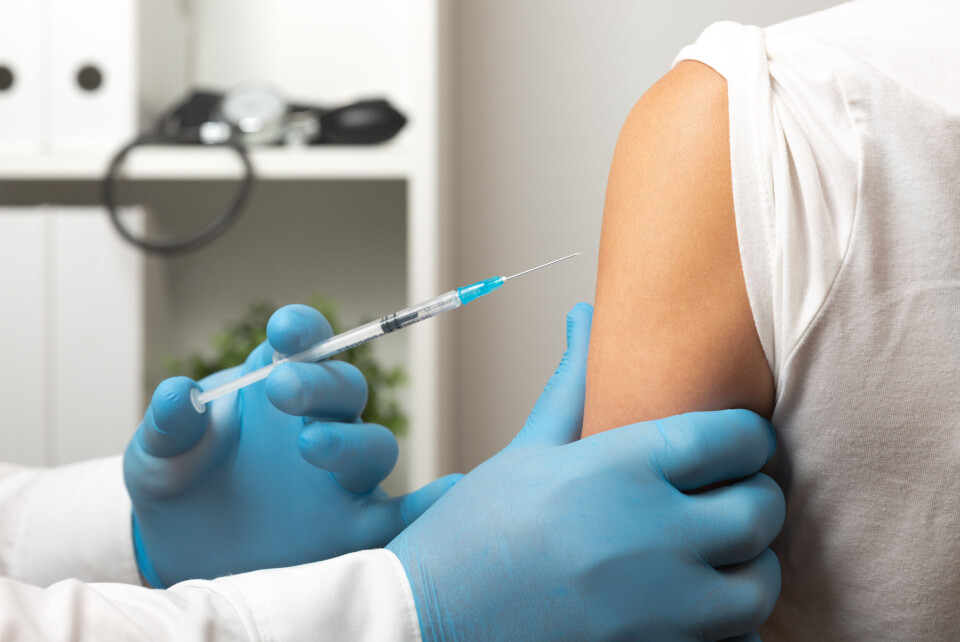
A French company is presenting its groundbreaking invention – a cancer ‘vaccine’ that limits the recurrence of the disease – at the annual congress of the American Society of Clinical Oncology (ASCO) in Chicago on Monday (June 5)
Targeted at those who have already suffered from a form of cancer, the vaccine uses genetic information from a patient’s tumour after it has been removed to limit the re-emergence of cancerous growths.
Transgene, based in Strasbourg, has developed the jab primarily for ear, nose, and throat (ENT) cancers, but has other products in the works to combat other forms of the illness.
In the first trial of the ENT vaccine, all 16 patients who received it successfully avoided a relapse.
Although this was only the first phase of a number of trials needed before the vaccine can be made commercially available, the team are confident of its success.
Prevents cancer from returning
Despite being listed as a cancer ‘vaccine’, the jab in question is not a preventative to lower the risk of getting cancer for the first time, but one to prevent cancer from recurring in an already-sick patient.
The choice to develop the vaccine for ENT cancer sufferers is because it has some of the highest levels of relapse.
The jabs are developed individually for each patient at the company’s lab, using genetic material gathered from a patient’s tumour post-operation.
An Artificial Intelligence (AI) machine then studies this information to predict what 30 cancerous mutations (out of a possible 3,000) are most likely to appear for the patient.
Like a regular vaccine, the information embedded within the jab helps to ‘teach’ the body to fight against the emergence of the 30 chosen mutations by using antibodies produced by the patient’s immune system.
“It’s like finding a needle in a haystack,” said Transgene’s CEO Hedi Ben Brahim, before adding the use of AI is necessary to “detect the best mutations to make a truly effective vaccine.”
The jabs are then created at the company’s labs, before being given to patients in 20 overall doses.
Read more: French attitudes to smoking: Six of the latest trends
Future plans
The team will be present at ASCO’s annual congress to present the success of the first human trials, but this is only a preliminary step in the vaccine’s progress.
After this, further phases of testing will see hundreds, and eventually thousands, of patients be tested with the vaccine before it can be made available to the wider public.
Alongside the ENT vaccine, the company is also looking at developing a vaccine for the Papillomavirus (HPV).
























